Barabino Silvia, PhD
Associate Professor of Molecular Biology

room 3049, building U3, tel.: +39 02 6448 3352
lab 3048, building U3, tel. +39 02 6448 3348
Group leader - Biologia dell'RNA
Area di ricerca: BIOMEDICINA
Keywords
gene expression; RNA processing; DNA damage; RNA-bindig proteins; human disease neurodegeneration
Graphical abstract
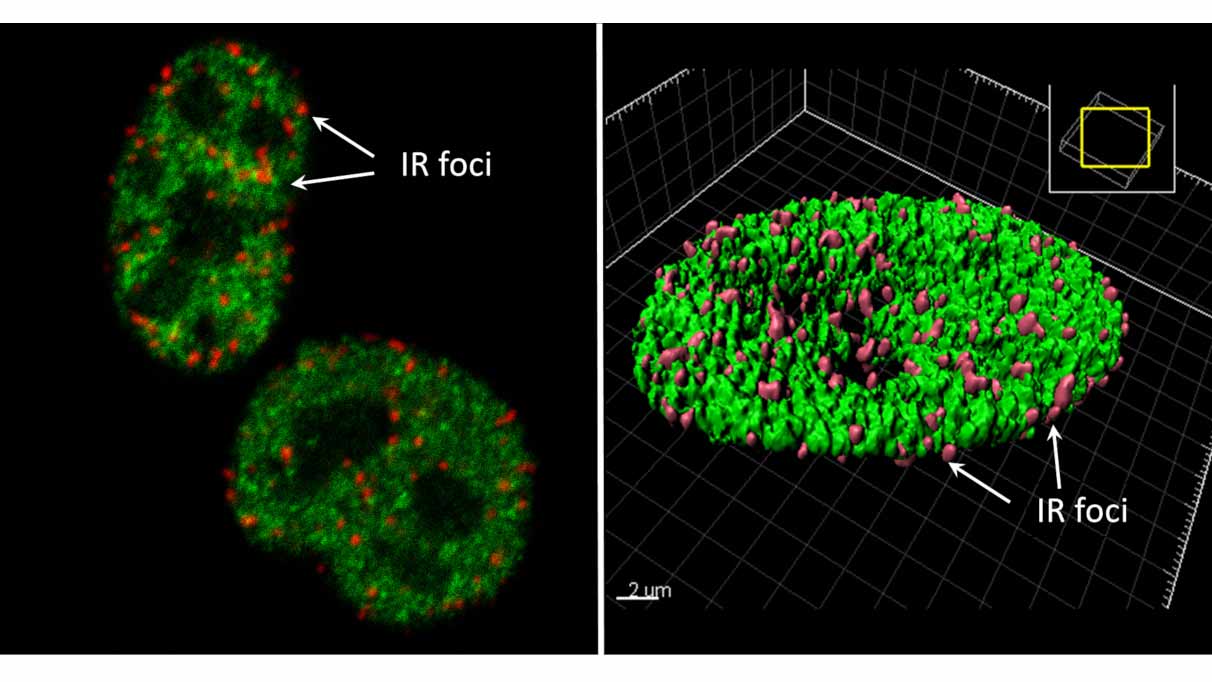
Research interest
Our research activity is focused on the investigation of the role of RNA-binding proteins in transcription and RNA metabolism and how these mechanisms contribute to human diseases, with a specific focus on neurodegeneration. Currently, we are concentrating on two main lines of research. The first one is focused the characterization of miRNA expression in cellular and mouse models of Amyotrophic Lateral Sclerosis. The second line relates to on the characterization of the role of in genome stability in neurodegeneration. Specifically we are currently investigating the role of the FET family of RNA-binding proteins in the DNA damage response and how mutations I these proteins affect genome stability.
To address these questions, we use a combination of biochemical and molecular approaches, microscopy, in vitro cell culture systems, and transgenic mouse models.
Research projects
- The function of FET proteins in genome stability as a pathogenic pathway for ALS/FTD
- Targeting miR129-1 as therapy for sALS and fALS
Selected articles
-Loffreda A, Nizzardo M, Arosio A, Ruepp MD, Calogero RA, Volinia S, Galasso M, Bendotti C, Ferrarese C, Lunetta C, Rizzuti M, Ronchi AE, Mühlemann O, Tremolizzo L, Corti S, Barabino SML. miR-129-5p: A key factor and therapeutic target in amyotrophic lateral sclerosis. Prog Neurobiol. 2020 Jul;190:101803. doi: 10.1016/j.pneurobio.2020.101803.
-Barbarani G, Fugazza C, Barabino SML, Ronchi AE. SOX6 blocks the proliferation of BCR-ABL1+ and JAK2V617F+ leukemic cells. Sci Rep. 2019 Mar 4;9(1):3388. doi: 10.1038/s41598-019-39926-4.
-Fontana GA, Rigamonti A, Lenzken SC, Filosa G, Alvarez R, Calogero R, Bianchi ME, Barabino SM. Oxidative stress controls the choice of alternative last exons via a Brahma-BRCA1-CstF pathway. Nucleic Acids Res. 2017 Jan 25;45(2):902-914. doi: 10.1093/nar/gkw780.
-Reber S, Stettler J, Filosa G, Colombo M, Jutzi D, Lenzken SC, Schweingruber C, Bruggmann R, Bachi A, Barabino SM, Mühlemann O, Ruepp MD. Minor intron splicing is regulated by FUS and affected by ALS-associated FUS mutants. EMBO J. 2016 Jul 15;35(14):1504-21. doi: 10.15252/embj.201593791.
International and national collaborations
- Marc-David Ruepp, UK Dementia Research Institute at King's College London, U.K.
- Oliver Muehlemann, Department of Chemistry and Biochemistry, University of Bern, Switzerland.
- Neus Visa, Department of Molecular Biosciences, The Wenner-Gren Institute, Stockholm University, Stockholm, Sweden.
- Stefania Corti, Dino Ferrari Centre, Neuroscience Section, Department of Pathophysiology and Transplantation (DEPT), University of Milan, Italy
- Emanuele Buratti, International Centre for Genetic Engineering and Biotechnology (ICGEB), Trieste, Italy
Barabino’s Lab – #BarabinoLab_BtBS
last update October 2020